Nov 02 2015
Nerve Stimulation for Relaxation
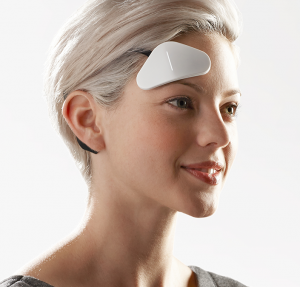 A new device called Thync has been advertising quite extensively and this has prompted a number of questions to me about its legitimacy. The device is worn over the right temple and above the right eye and provides timed pulses of mild electrical stimulation. Their website describes the effects:
A new device called Thync has been advertising quite extensively and this has prompted a number of questions to me about its legitimacy. The device is worn over the right temple and above the right eye and provides timed pulses of mild electrical stimulation. Their website describes the effects:
A soothing neck massage. A splash of cold water. A kiss from someone you love. These are common examples of how nerves signal the brain to change the way you feel. Thync works in the same way using signaling programs we call Vibes.
The device appears to have two settings, one with calming vibes and the other with energizing vibes. Let’s take a look at the state of the science
Plausibility
The idea of using electrical stimulation, of either nerves, the skin, or of muscles, in order to affect nervous system function is reasonable. The nervous system is both a chemical and electrical organ system and its function can be modified with electrical stimulation.
Current applications include using transcutaneous electrical nerve stimulation (TENS) to reduce pain. The concept of TENS is to stimulate nerves with non-painful stimuli in order to inhibit the conduction of painful signals. A recent review of 19 clinical trials of TENS in acute pain found that there may be an effect above placebo, but the data is still preliminary. A separate review of TENS for central pain in multiple sclerosis was a little more positive, concluding there is level 2 evidence to support efficacy (the gold standard is level 1 evidence).
Overall the evidence suggests that TENS is moderately effective for pain, but the data is still a bit unclear.
Electrical skin stimulation is also being studied for acute migraine attacks, and there is one FDA approved device on the market. However, the data for this treatment is very preliminary, and I was actually a bit surprised that the FDA approved the device with such evidence.
Research is also underway looking at trigeminal nerve stimulation for depression. A recent pilot study found some effects, but this was an open-label study of only 11 subjects. This was presented as a “proof of concept” only, in order to justify further research. A 2014 review of deep brain stimulation, transcranial magnetic stimulation, and trigeminal nerve stimulation for depression concluded, essentially, that more research is needed.
I won’t further consider deep brain stimulation or transcranial magnetic stimulation here as their relevance is limited, in my opinion, because these treatments stimulate the brain directly rather than through peripheral nerves.
What we can say at the present time is that using electrical stimulation of one kind or another to modulate brain function is a plausible approach with some early indications. Direct central stimulation is better established than peripheral stimulation, which remains more experimental. Effects of peripheral stimulation on pain, migraine, and mood appear to be modest and the data is all preliminary. It is not yet clear if the limited effects seen are a result of limitations to this approach, or of the current state of the technology. We therefore don’t know what the ultimate potential will be.
The Evidence
The two claims being made for Thync, that it can stimulate the trigeminal nerve to produce either relaxation or stimulation, are not implausible, but what is the current state of the evidence? The company claims:
World-class neuroscience has been present from day one and forms the core investment in our company.
They also provide links to “Thync Science.” It is great when a company does the leg work and compiles all the relevant scientific evidence in support of their claims. Why would they not do this? Unfortunately, what often happens is that companies link to a bunch of studies that are only tangentially related to their claims, or even have absolutely nothing to do with their claims. It is also a safe bet that they are cherry picking the studies they present to support their claims. So you can never rely upon what the company presents.
However – it is still extremely useful. Making a negative statement, that no study exists to support a claim, for example, is always tricky because it means you did a really thorough search. Without getting into detail, there are lots of places for studies to hide, and searching is not always straightforward. If a company lists supporting research, however, then it is a pretty safe bet that if a solid supporting study existed, they would list it. Of course, I always do my own search as well.
In this case the company lists, and I can only find, one clinical study that can be used to support their claims. The other listed studies are either not directly related to the technology (the researchers, for example, also did studies using ultrasound stimulation) or are not testing clinical claims.
Here is the one relevant study, which tests the relaxation effects of Thync. I could find no studies at all testing the stimulation claims for the device.
The study looks at markers of sympathetic response and subjective reporting of relaxation, in a blinded comparison between Thync stimulation and active sham stimulation. They report statistically significant effects of Thync over Sham.
The text of the report reads like an industry-insider study. While it is reasonably scientific, the bias of the researchers is evident. They also disclose at the end:
WJT, JDC, DZW, and SKP are inventors and coinventors on issued and pending patents related to methods, systems, and devices for noinvasive neuromodulation. All authors are shareholding employees of Thync, Inc.
All the authors therefore have a direct massive conflict of interest, and we know that such conflicts affect the outcome of clinical trials.
The study itself was fairly small, with only 87 subjects. While the comparisons were double-blinded, there is no indication that the blinding was assessed in any way. This is a significant shortcoming, as the research in general with electrical stimulation has a problem maintaining blinding. Failing to assess blinding, by itself, might be a fatal flaw.
Further, in the part of the experiment where they assess subjective relaxation the description indicates that every subject received both sham and active treatment and their responses were compared. No mention is made of whether or not the order in which the treatments were given was randomized and balanced. This is a significant oversight. Either they didn’t randomize the order, or they didn’t think it was worth mentioning in the description of their methods.
This is a major problem, because if the sham treatment, for example, was always given first then that might compromise blinding. Further, there may have simply been a cumulative placebo effect, with the second treatment always being rated better.
While the results were statistically significant, the confidence intervals overlap (sham = 2.82 ± 2.03, TEN = 5.15 ± 2.14; p = 2.63 x 10-11; Figure 3A). That means, within the 95% confidence interval, the effects might have been the same. The effects were therefore not that statistically impressive, despite the low p-value.
Conclusion
I do think we are heading into an era of using various kinds of both electrical and magnetic stimulation as a therapeutic tool. The low hanging fruit are probably pain (including migraine) and depression, but other applications are possible also. Vagus and deep brain stimulation are being used for things like seizures and tremors as well.
We are relatively near the beginning of this technology, however. It takes years, even decades, and many studies to sort out exactly what works and what doesn’t. At the very least (just like with drugs) I would like to see several large rigorous clinical trials showing a clear effect before a product can be marketed with specific claims.
The Thync device, in my opinion, is jumping the gun and making claims that go beyond the evidence. All they currently have is one small in-house study looking at one of their two claims (relaxation). Also, using one device stimulating at the same location for both relaxation and stimulation stretches plausibility. I can’t say it’s impossible, because the intensity and frequency of stimulation does matter, at least with transcranial magnetic stimulation, but I don’t see any evidence to support the claim that peripheral simulation can have essentially opposite effects.
The Cephaly device for migraine actually has FDA approval, but that is because they are making a disease claim (migraine). I don’t see any mention on the Thync website of the FDA, probably because they are not making disease claims but “structure function” claims. Also, I don’t see the standard disclaimer that their claims have not been evaluated by the FDA.
Overall, at this point I would advise caution. The evidence just does not seem to be there for the specific claims being made for this device.






